Executive Summary
The 2026 JPMorgan Healthcare Conference made one thing unmistakably clear: rare-disease companies are no longer judged solely on scientific novelty. Investors are demanding commercial maturity, operational discipline, and evidence-ready platforms—long before approval. Capital is returning to the sector, but it is flowing selectively to companies that demonstrate a credible path to revenue and the infrastructure to scale.
Below we outline the five imperatives rare-disease companies must emphasize in their 2026 fundraising narratives and provide a practical Commercial Readiness Scorecard to help leaders benchmark their preparedness.
1. Demonstrating Commercial Maturity—Earlier Than Ever
The historical model—raise capital on science, build commercial capabilities later—is no longer viable. Investors now expect rare-disease companies to behave like mature biopharma organizations well before launch.
Signals investors look for:
- A defined launch readiness framework
- Early payer and KOL engagement
- Patient-finding and diagnostic strategies
- Cross-functional governance and decision-making discipline
- Clear articulation of global expansion pathways
In rare disease companies, commercial readiness is not a late-stage activity—it is a risk-mitigation strategy. Companies that can show operational foresight are perceived as lower-risk investments with higher probability of launch success.
2. Articulating a Credible Path to Revenue
With capital still constrained, investors are prioritizing assets with near-term value inflection and clear regulatory trajectories. Rare-disease companies must demonstrate how they will convert scientific promise into revenue.
Key elements of a credible revenue narrative:
- Clear timelines to approval and launch
- Early market and expanded access strategies
- Pricing and reimbursement assumptions grounded in analogs
- Market development plans for ultra-rare populations
- Evidence that the company understands payer skepticism and has a plan to address it
Investors want to see not just when revenue will arrive, but how the company will de-risk the path to it.
3. Differentiated Clinical and Mechanistic Storytelling
In a crowded rare-disease landscape, differentiation is currency. Companies must articulate why their asset is not only scientifically compelling but commercially meaningful.
What strong differentiation looks like:
- A mechanistic rationale tied to disease biology
- Clinically meaningful endpoints aligned with regulatory and payer expectations
- A biomarker or diagnostic strategy that strengthens the value story
- Real-world evidence plans that extend beyond approval
The most compelling narratives connect mechanism, clinical design, and commercial value into a single, coherent story.
4. Operational Discipline and Focused Investment
Investors are rewarding companies that demonstrate discipline—lean cost structures, focused pipelines, and clear prioritization.
Signals of operational excellence:
- A right-sized organization with clear accountability
- Early, disciplined spend on CMC, evidence generation, and market development
- A pipeline strategy that balances optionality with focus
- Early manufacturing readiness, especially for gene and cell therapies
Rare-disease companies that show they can operate with precision and efficiency stand out in a capital-selective environment.
5. Optionality and Strategic Flexibility
The most attractive companies are those that create multiple paths to value creation. Investors want to see optionality—strategic partnerships, platform leverage, and business development-ready data packages.
Optionality can include:
- Multiple shots on goal within a platform
- Clear partnering strategies for ex-US markets
- BD-ready data packages aligned with pharma expectations
- AI-enabled efficiencies in patient identification, trial recruitment, and evidence generation
Optionality signals resilience. It shows investors that the company can create value even if the primary path encounters friction.
Commercial Readiness Scorecard
Below is a detailed Commercial Readiness Scorecard designed for rare disease companies to benchmark their preparedness across key investor-focused dimensions. This scorecard can be used as a practical tool to assess commercial maturity and readiness for fundraising and launch success.
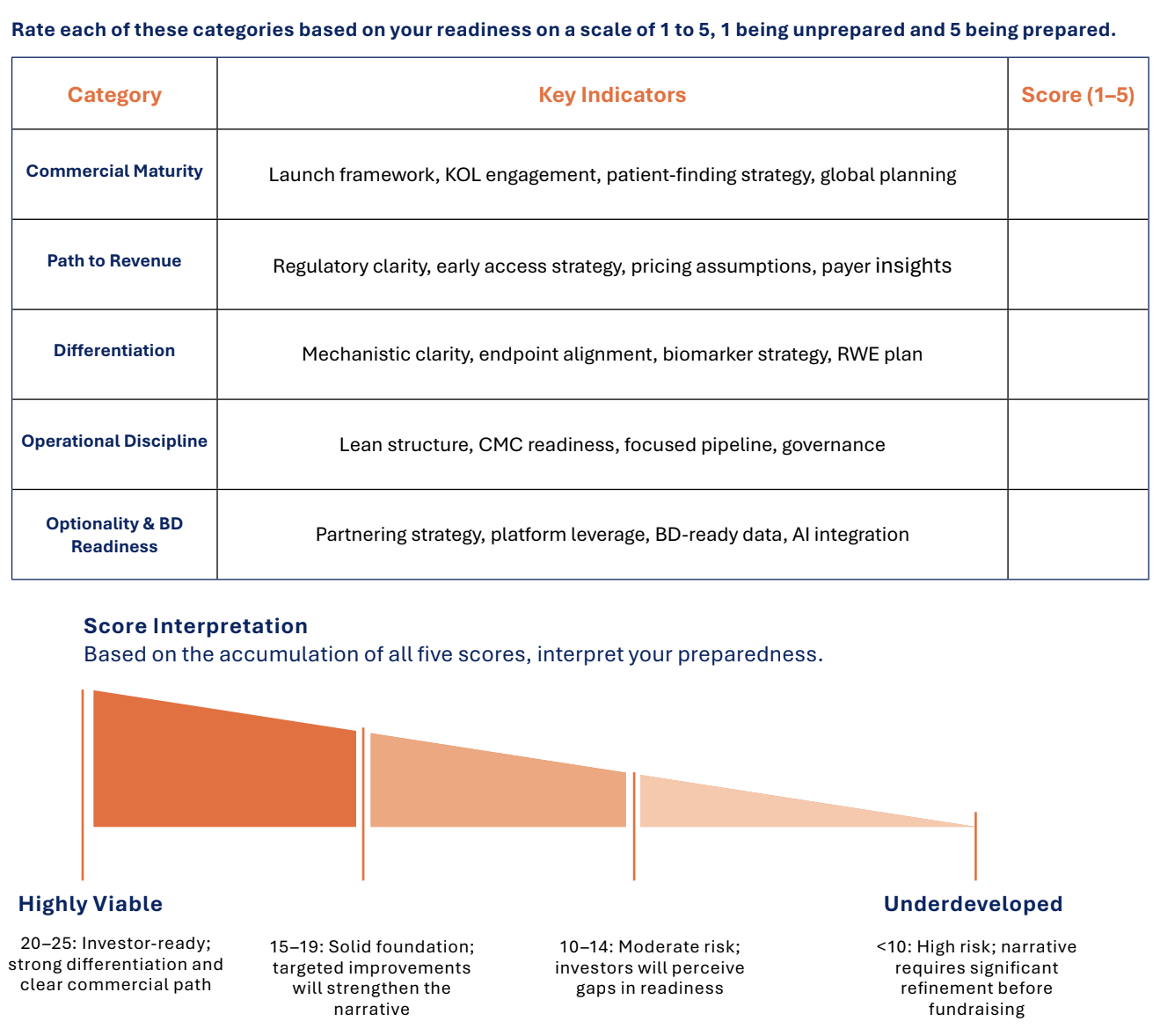
This scorecard is intended to be used as a practical guide for leadership teams to evaluate their commercial readiness and identify areas for strategic focus and improvement.
Conclusion: The Rare-Disease Leaders Who Win in 2026
The rare-disease companies that will stand out in 2026 are those that combine scientific excellence with commercial sophistication. Investors are no longer funding potential—they are funding preparedness. Rare-disease companies that can demonstrate maturity, discipline, and strategic flexibility will not only secure capital but accelerate their path to meaningful patient impact.
Are you ready to win as a Rare Disease leader in 2026? Reach out to Triangle Insights Group today.


